
Quality Risk Management (QRM) activities shall include systematic processes designed to coordinate, facilitate and improve science-based decision-making with respect to risk.Initiating a Quality Risk Management (QRM) process:.A typical quality risk management process is given below.The level of effort, formality and documentation of the Quality Risk Management (QRM) process should be commensurate with the level of risk.The evaluation of the risk to quality should be based on scientific knowledge and ultimately linked to the protection of the patient.The two primary principles of Quality Risk Management (QRM) are that:.Principles of Quality Risk Management (QRM):.Additional info, including the proposed mitigation or actions used to lower a risk or justify a risk level or scenario. 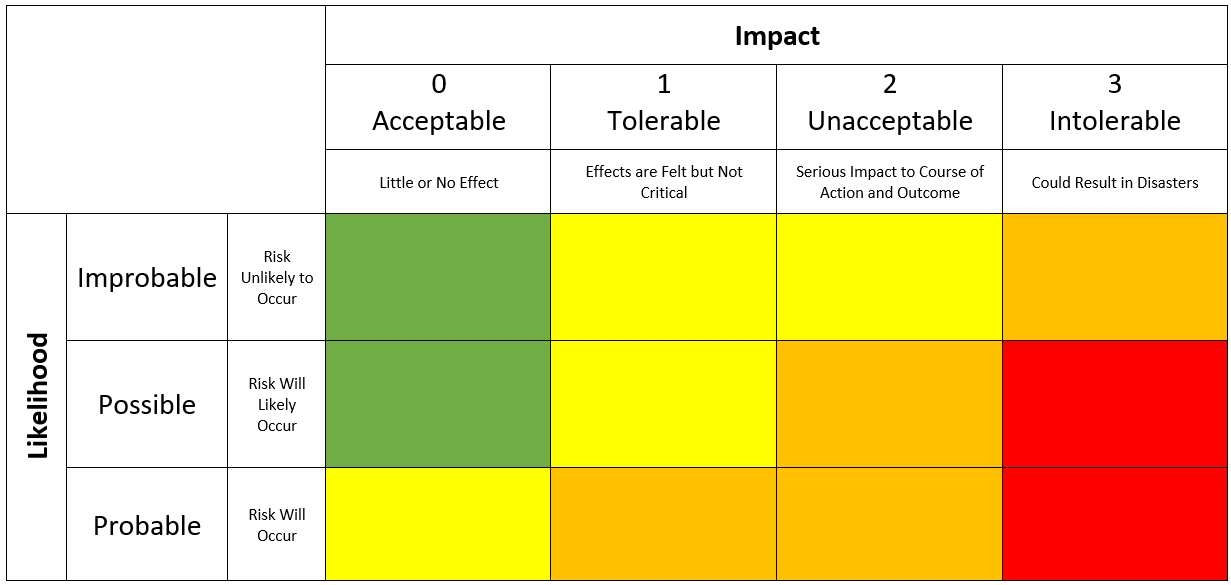
A measure of the possible consequences of a hazard.The assumption is that the cause has occurred.It is a numerical subjective estimate of the effectiveness of the controls to prevent or detect the cause or failure mode before the failure reaches the customer.The means of detection of the failure mode by maintainer, operator or built in detection system, including estimated dormancy period (if applicable) or it is also sometimes termed EFFECTIVENESS.Review or monitoring of output or results of the risk management process considering (if appropriate) new knowledge and experience about the risk.RPN = severity rating × occurrence rating × detection rating. The product of these three scores is the RPN for that failure mode.Each failure mode gets a numeric score that quantifies likelihood of occurrence, likelihood of detection and severity of impact.A numeric assessment of risk assigned to a process, or steps in a process, as part of failure mode effects and criticality analysis (FMECA).The systematic use of information to identify potential sources of harm (hazards) referring to the risk question or problem description.The comparison of the estimated risk to given risk criteria using a quantitative or qualitative scale to determine the significance of the risk.The sharing of information about risk and risk management between the decision maker and other stakeholders.It consists of the identification of hazards and the evaluation of risk associated with exposure to those hazards.A systematic process of organizing information to support a risk decision to be made within a risk management process.The estimation of the risk associated with the identified hazards.Risk: Combination of the probability of occurrence of harm and severity of the harm.A systematic process for the assessment, control communication, and review of risks to the quality of the pharmaceutical product across the product life-cycle.Probability of negative events within a fixed time frame.Different ways that a process or sub-process can fail to provide the anticipated result.A process parameter which could have an impact on the critical quality attribute.A physical, chemical, biological or microbiological property or characteristic that should be within an appropriate limit, range, or distribution to ensure the desired product quality.A systematic method of identifying and preventing product and process problems.Failure Mode, Effects And Criticality Analysis (FMECA):.Responsible for effective implementation of this SOP.Ĥ.0 Procedure: Quality Risk Management (QRM) : Responsible for approval of documents after analysis and conclusion. Responsible for identification of risk and communication of risk to Quality Risk Management (QRM) team.įormation of Quality Risk Management Team and team leader. Technical staff/ Officer/ Executive/ Manager – All departments Responsible for assigning Risk Assessment Document No. Preparation of action plan in case of higher RPN and communication to Head – QA, department Head. Identifying all perceived failures with respect to process, equipment, facilities and system. This guideline is applicable to identify the quality risks involved in any process, equipment, facilities and system.ģ.0 Responsibility Quality Risk Management (QRM) Team.To lay down the procedure for risk identification, analysis, evaluation, reduction/ mitigation, communication and conclusion of risk in order to ensure the quality, safety, integrity and purity of the drug product.Quality Risk Management (QRM) 1.0 Objective :
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
